Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Arisaka, Makoto*; Kimura, Takaumi; Suganuma, Hideo*; Yoshida, Zenko
Radiochimica Acta, 89(8), p.593 - 598, 2001/08
Times Cited Count:11 Percentile:61.93(Chemistry, Inorganic & Nuclear)no abstracts in English
Makuuchi, Keizo
Porima Daijesuto, 51(12), p.84 - 104, 1999/12
no abstracts in English
Lothenbach, B.*; Ochs, M.*; Wanner, H.*; Yui, Mikazu
JNC TN8400 99-011, 340 Pages, 1999/01
This report provides thermodynamic data for predicting concentrations of palladium Pd, lead Pb, tin Sn, antimony Sb, niobium Nb and bismuth Bi in geologic environments, and contributes to an integration of the JNC chemical thermodynamic database, JNC-TDB (previously PNC-TDB), for the performance analysis of geological isolation system of high-level radioactive wastes. Besides treating hydrolysis in detail, this report focuses on the formation of complexes or compounds with chloride, fluoride, carbonate, nitrate, sulfate and phosphate. Other important inorganic ligands (sulfide for lead and antimony, ammonia for palladium) are also included. In this study, the specific ion interaction theory (SIT) approach is used to extrapolate thermodynamic constants to zero ionic strength at 25 C.
C.
Kimura, Takaumi; Kato, Yoshiharu; Takeishi, Hideyo; G.R.Choppin*
Journal of Alloys and Compounds, 271-273, p.719 - 722, 1998/00
Times Cited Count:48 Percentile:88.86(Chemistry, Physical)no abstracts in English
; Kaetsu, Isao
Int.J.Appl.Radiat.Isot., 35(1), p.21 - 24, 1984/00
Times Cited Count:6 Percentile:57.37(Nuclear Science & Technology)no abstracts in English
Umezawa, Hirokazu; ;
JAERI-M 5785, 11 Pages, 1974/07
no abstracts in English
; Ono, Shinichi;
Radioisotopes, 21(8), p.478 - 481, 1972/08
no abstracts in English
*; *; *; *
Nihon Genshiryoku Gakkai-Shi, 5(7), p.566 - 571, 1963/00
no abstracts in English
Sasaki, Yuji; Kaneko, Masashi; Ban, Yasutoshi; Kinoshita, Ryoma*; Matsumiya, Masahiko*; Shinoku, Kota*; Shiroishi, Hidenobu*
no journal, ,
We obtained relatively high D(Rh) of approximate 1 by iminodioctamide (IDOA) having tertially amino N atom from concentrated HCl solution. The reason is that IDOA is protonated, behaves as cationic extractant, and extracts anionic RhCl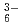 through ion-pair extraction. Up to now, there is less information on Rh(III) extraction, we investigate the behavior of Rh extraction and discuss with theoretical studies.
through ion-pair extraction. Up to now, there is less information on Rh(III) extraction, we investigate the behavior of Rh extraction and discuss with theoretical studies.
Kinoshita, Ryoma; Sasaki, Yuji; Kaneko, Masashi; Matsumiya, Masahiko*; Shinoku, Kota*; Shiroishi, Hidenobu*
no journal, ,
Many metal ions are stable in hydrochloric acid solutions as anionic species; diamidic-extractants containing tertiary amino nitrogen atom, such as NTAamide (hexaalkyl-nitrilotriacetamide), are normally protonated in acidic solutions and become cationic extractants. In this study, the ion-pair extraction reactions of metal chloride anions with cationic NTAamide(C6) extractant were investigated. In addition, we attempted to predict the distribution ratio by combining the stability constants of metal chloride complexes and DFT calculations, and compared the results with experimental values.
Sasaki, Yuji; Ban, Yasutoshi; Matsumiya, Masahiko*
no journal, ,
Metal ions are present as anionic species in hydrochloric acid. Diamino-extractants containing tertiary amino N atom, such as NTAamide, protonate in acidic solutions and become cationic extractants. In this study, the ion-pair extraction of metal chloride anions, especially platinum metals and the related metals, with the cationic extractants was performed and their extraction properties are studied.